By Carla M. Quiñones
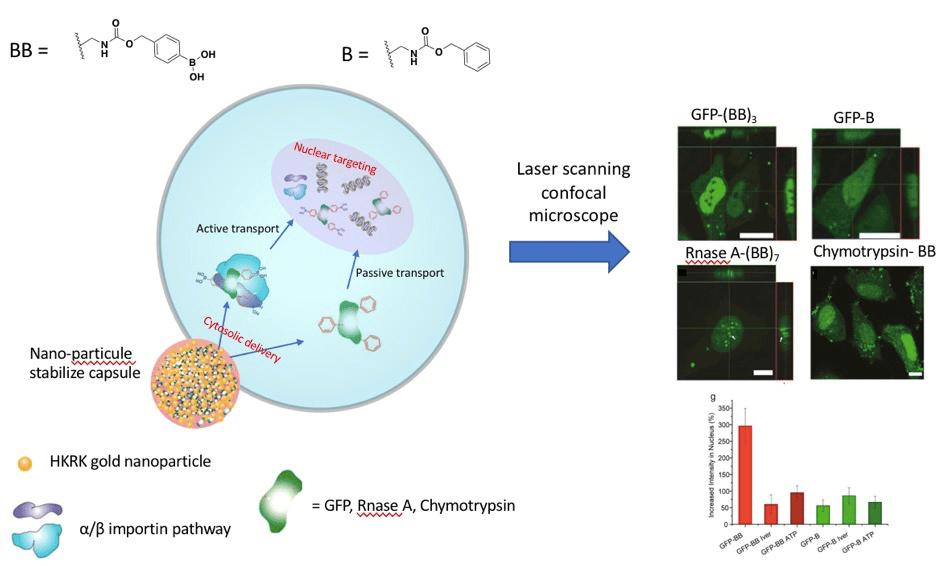
The main objective of the research described in this article by Rotello and coworkers was to use a synthetic non-peptide targeting motif that accesses the nucleus of the cell through an active transport mechanism. Active and passive transport take place in the cellular membrane as well as in the nuclear membrane. Intracellular targeting is as important as cellular targeting due to their relevance in drug delivery and potential therapies. The two challenges faced by the research group was to deliver the nuclear-targeted protein into the cytosol and to comprehend the mode of nuclear entry. They delivered the protein successfully to the cytosol with the help of a previously synthesised nanoparticle stabilized particle, which encapsulated the proteins of interest and liberated them into the cytosol via membrane fusion. The most significant finding was the efficiency of targeting the nucleus when modifying each protein of interest with benzyl boronate tags (BB tags).
They performed experiments with different proteins modified with BB tags resulting in a successful high-efficiency delivery to the nucleus. To assess the role of boronic acid in the BB tags, they modified GFP with the benzyl tag alone and saw less fluorescence inside the nucleus. This suggests that the boronic acid is necessary in the BB tags for a successful nuclear targeting. Furthermore, they determined if the mechanism of transport into the nucleus was either active or passive. To accomplish this goal, they added Ivermectin, an inhibitor for the α/β importin pathway (active transport), to the cells and also depleted ATP (required for all active transport pathways) in another set of experiments. They delivered GFP with 3 BB tags in both cases and saw a lower nuclear efficiency. They concluded that these modified proteins targeted the nucleus effectively through the importin α/β pathway (active transport) rather than passive transport.
Through the last part of the paper, I was curious of how they acknowledged that this happened the way the proposed since they didn’t explain the mechanism behind the α/β importin pathway, nor the chemistry of how the BB tag contributes to the nuclear targeting. Intrigued by this, I searched more about this phenomenon, but found no relevant studies, probably because this is a novel research field. In general, this was a good article and useful to our lab since we work with supramolecular systems which, in the future, we could adapt this strategy to achieve nuclear targeting.