By Luis A. Prieto
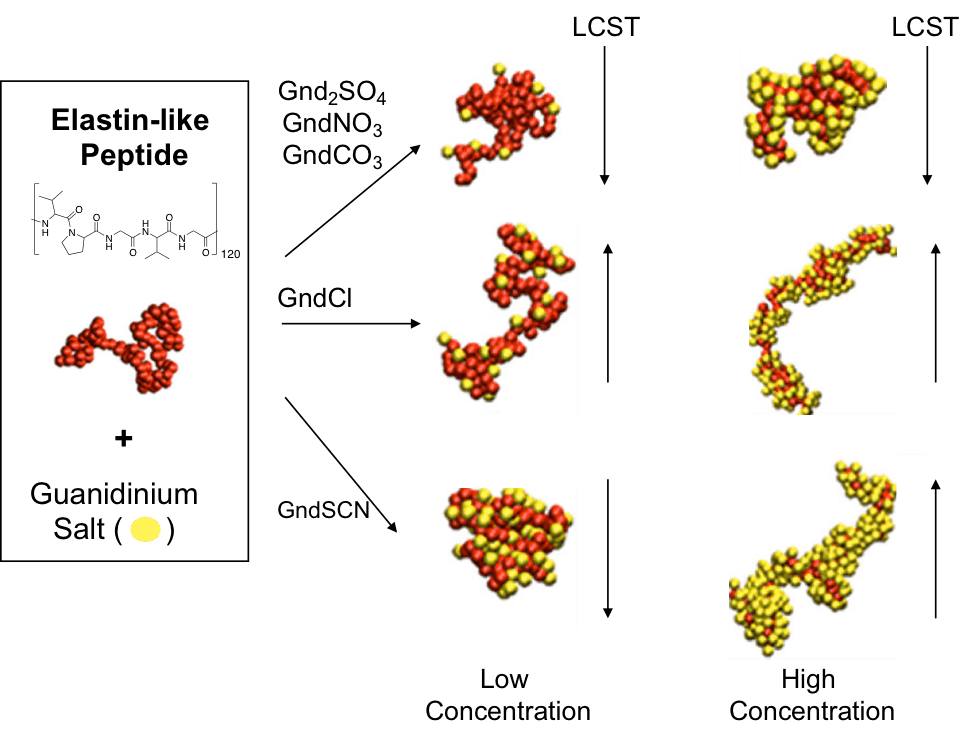
Heyda, Jungwirth and Cremer collaborated in a study of guanidinium (Gnd+) salts and their effect in lower critical solution temperature (LCST). They studied molecular details of the cause of these transitions by IR-ATR and molecular dynamics simulation where they wanted to understand how Gnd+ salts interacted with the backbone of the Elastin-like peptides (ELP). In previous studies ELPs showed a change in LCST that followed the Hofmeister series in sodium salts but using Gnd+ salts proved to be different, especially Guanidinium thiocyanate (GndSCN) that at low concentrations the LCST decreases, but at high concentrations the LCST increases. They studied particular phenomenon using ATR-IR where they found GndSCN binds strongly with ELPs and resulted in an interesting behavior when the concentration of salt is increased. At low concentrations the polymer collapsed (salting-out) because of cross-linking of the peptide and at high concentrations resolubilization occurred (salting-in). Other salts followed typical behavior of salting-in (guanidinium chloride, weak binding) or salting-out (guanidinium sulfate, poor binding). Coarse-grain and all atom simulations corroborated this finding where they found particular detail of the interaction of the carbonyl groups of the peptide backbone with Gnd+, most likely through H-bonds.
The thermodynamics of this paper I found particularly interesting since it reminded me of everything that I have to re-learn. An attractive experiment was that they used a melting point apparatus to measure the LCST, meaning the use of small amount of sample to gather fundamental information of the system which is also the case with ATR-IR. An elegant work and also inspirational since our lab works with responsive systems and we will definitively see if we can do the LCST measurement with a melting point instrument. I got to say that I particularly like the all atom simulations and Figure 5, where we can see in molecular detail the interactions of the salts with the peptide where thiocyanate and Gnd+ interact strongly with the hydrophobic parts (V, G) and hydrophilic part (peptide bond), respectively.
LAPC: Heyda, 2017. Guanidinium can both Cause and Prevent the Hydrophobic Collapse of Biomacromolecules
Pingback: Maxier’s comment on: Macromolecular Crowding Modifies the Impact of Specific Hofmeister Ions on the Coil–Globule Transition of PNIPAM – Rivera Lab at UPR
Rating (synopsis): 4/5
Rating (figure): 5/5
A surprisingly short but satisfying synopsis. The gist of the paper is given while avoiding almost all detail. Very Prieto-like. I think I’d have included a few more take-home messages from the paper, since the most important results aren’t the observed fact but rather the explanations, which are a lot more elaborate in the paper. However, that is most likely a matter of style: I’m a lot more wordy than Prieto.
A few minor errors in writing in acronyms (IR-ATR vs ATR-IR), redundancy (…corroborated this finding where they found…) or a little Yoda-like style (…Guanidinium thiocyanate (GndSCN) that at low concentrations the LCST decreases, but at high concentrations the LCST increases). But overall, well-written.
The paper is more clearly understood through images than description. I really liked the synopsis figure, even more than the TOC image from the original paper. I might have added the graph from figure 2, which I think helps understand (or reminds us of) the results quicker.
Rating (synopsis): 5/5
Rating (figure): 4/5
I liked the summary of Luis since it was direct and with a simple vocabulary. In the presentation I was a little bit confuse so I didn’t understand the article but with its summary I could understand the main purpose of the article. Again, using techniques that I am related and I like that because it helps me to understand more what they are doing in their research.
The image is constructive and simple. Explain the main experiment of the article and which had the greater focus that was the modulation of LCST.
Rating (synopsis): 5/5
Rating (figure): 4.5/5
Since the presentation I’ve been very impressed by this paper and how complicted it is. Prieto’s understanding of it is shown through his synopsis. He takes a ton of results and simplifies them in a way that even undergrads can understand. However, it is still a synopsis, not every result and explanation has to be here, and deciding which one gets overlooked is also a sign of comprehension. I really like his comment at the end which was his take home message of applying techniques and instruments from the paper to our lab and his research.
The figure is serviceable, clean and well put together. Using clear imagery he summarized the majority of results. I wanted a little more creativity from Prieto so I will refrain from giving a perfect score, but apart from that the figure is great.
Synopsis: 5/5
Figure: 4/5
Prieto’s take on this paper is very on point without a lot ornaments or tangents. Tangents that while trying to give background, I catch myself going to when writing. Therefore, I can admire his take and comments without missing the point of the paper. The paper itself has a lot of detail and takes in consideration various experiments yet Prieto’s comment was useful to focus on what is important for us of this paper. His image, as his writing very “to the point”. Sadly he does not use the best organization of images to make clear the LCST changes. Here, some more attention to detail might have helped.
Synopsis: 5/5
Figure: 5/5
It was interesting to see how Luis was able to summarize the important discoveries of the paper in a short synopsis. In the figure, the main goal of the paper is clear, which is to ”study how the different guanidinium (Gnd+) salts influence the stability of the collapsed versus uncollapsed state of an elastin-like polypeptides (ELPs)”. I think it’s a very nice figure that explains what happens to the LCST when the Gnd+ salts are added to the peptide.
Rating (Synopsis): 4/5
Rating (Figure): 5/5
Given that the article was pretty dense and hard to understand, I am very fond of how Luis was able to present the main idea and results of the article in a very organized and simplified manner with this synopsis. I am very impressed by Luis’ ability to summarize the results, including how Gnd+ influences the stability of polypeptides under the circumstances presented in the paper, in such a short and concise synopsis (I know this is something very hard to do because the main issue I found while writing the synopsis of my paper was making it as short and concise as possible), and that needs to be appreciated. The only reason I’m giving this synopsis a 4/5 is that it has a few orthographical and grammatical errors that keep it from being perfect.
I prefer the TOC figure constructed by Luis much more than the one in the article because Luis’ figure summarized the results much better than the actual TOC figure. The article’s TOC figure seems much more complex than Luis’ figure.