By Luis A. Prieto
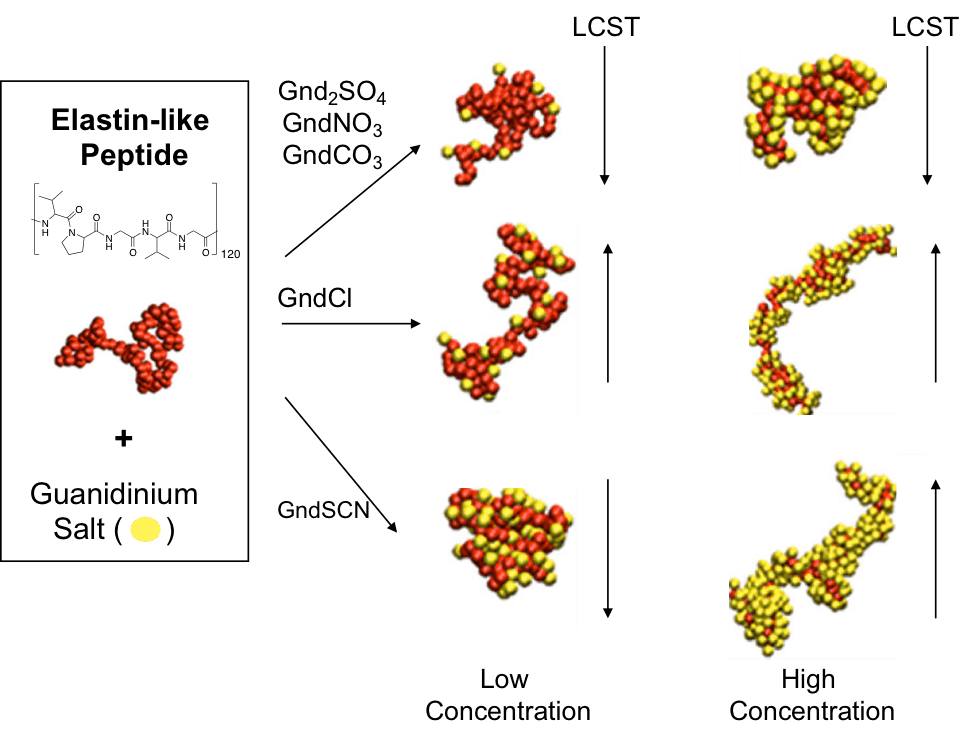
Heyda, Jungwirth and Cremer collaborated in a study of guanidinium (Gnd+) salts and their effect in lower critical solution temperature (LCST). They studied molecular details of the cause of these transitions by IR-ATR and molecular dynamics simulation where they wanted to understand how Gnd+ salts interacted with the backbone of the Elastin-like peptides (ELP). In previous studies ELPs showed a change in LCST that followed the Hofmeister series in sodium salts but using Gnd+ salts proved to be different, especially Guanidinium thiocyanate (GndSCN) that at low concentrations the LCST decreases, but at high concentrations the LCST increases. They studied particular phenomenon using ATR-IR where they found GndSCN binds strongly with ELPs and resulted in an interesting behavior when the concentration of salt is increased. At low concentrations the polymer collapsed (salting-out) because of cross-linking of the peptide and at high concentrations resolubilization occurred (salting-in). Other salts followed typical behavior of salting-in (guanidinium chloride, weak binding) or salting-out (guanidinium sulfate, poor binding). Coarse-grain and all atom simulations corroborated this finding where they found particular detail of the interaction of the carbonyl groups of the peptide backbone with Gnd+, most likely through H-bonds.
The thermodynamics of this paper I found particularly interesting since it reminded me of everything that I have to re-learn. An attractive experiment was that they used a melting point apparatus to measure the LCST, meaning the use of small amount of sample to gather fundamental information of the system which is also the case with ATR-IR. An elegant work and also inspirational since our lab works with responsive systems and we will definitively see if we can do the LCST measurement with a melting point instrument. I got to say that I particularly like the all atom simulations and Figure 5, where we can see in molecular detail the interactions of the salts with the peptide where thiocyanate and Gnd+ interact strongly with the hydrophobic parts (V, G) and hydrophilic part (peptide bond), respectively.
LAPC: Heyda, 2017. Guanidinium can both Cause and Prevent the Hydrophobic Collapse of Biomacromolecules