Month: June 2018
Rafa’s comment on: Thermal Switching of Thermoresponsive Polymer Aqueous Solutions
By Rafael A. Brito
Thermal switches are of great importance to thermal management in a wide variety of applications. A common characteristic associated with thermal switching is thermal conductivity. After noticing the change in thermal conductivity across LCST transitions, Zhiting Tian started researching polymers for this purpose. Poly(N-isopropylacrylamide) (PNIPAM) is the most studied thermoresponsive polymer and has a workable LCST of 32° C. The LCST transition of PNIPAM changes the chain-like formation of the polymer into an aggregation that shows a drastic decrease of thermal conductivity. This sharp change is due to LCST transitions being second-order, which are characterized by being almost instantaneous when the corresponding temperature is reached. Thermal conductivity was measured by applying a powerful approach: the transient thermal grating technique. It is used by heating a solution as a function of position creating a grating of temperature. This grating allows the use of the one-dimensional heat equation, which can be solved to give a relation between the thermal conductivity and temperature. The thermal conductivity can then be calculated using 𝑘 = 𝜌𝑐𝑝α, where 𝜌 is the density, is the specific heat capacity and α which is a function of temperature. After the setup was completed, solutions of varying concentrations were analyzed. For the solution with the highest concentration, the thermal switching ratio was measured to be 1.15 across the LCST transition. This shows a significant change between the two states of the polymer. The thermal conductivity of the PNIPAM aqueous solutions increases with temperature, the same as with water, until reaching the LCST. Then a drastic change is observed in the solution. The thermal switching ratio of PNIPAM aqueous solutions across the transition keeps increasing with increasing concentration, which is expected from the equation. To explain the thermal conductivity change due to the transition between the two modes of the polymer, the authors used the idea that the homogeneous phase of the solution separates into two phases that increases the thermal interface resistance resulting in a lower effective thermal conductivity.
As a summary, they reported the first direct measurement of thermal conductivity change in PNIPAM aqueous solutions across the LCST using a powerful approach, the laser-induced transient thermal grating technique. The results show an abrupt thermal conductivity drop across the transition temperature. The potential of using thermoresponsive polymer aqueous solutions of higher-order phase transitions for thermal switch applications has been demonstrated throughout this paper’s work.
Yanira’s comment on: Intracellular Guest Exchange between Dynamic Supramolecular Hosts
By Yanira Rodríguez Valdéz
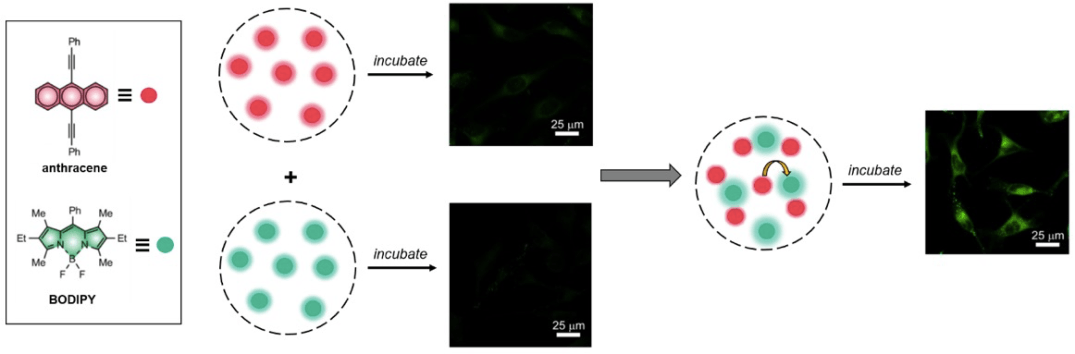
The article by Swaminathan et al. studies the Förster Resonance Energy Transfer (FRET) between two dyes encapsulated in self-assembled nanoparticles made from amphiphilic polymers. The encapsulation of hydrophobic chromophores such as borondipyrromethene (BODIPY) and anthracene inside the hydrophobic interior of these nanoparticles allows their study in aqueous media. In close proximity (inside the same host), BODIPY and anthracene undergo FRET (anthracene = donor, BODIPY = acceptor). The authors observed that mixing nanoparticles loaded with anthracene and nanoparticles loaded with BODIPY has the same effect as loading nanoparticles with both anthracene and BODIPY, which suggests guests being exchanged when nanoparticles are mixed and that they are being captured by the same nanoparticle(s). These studies are performed both in aqueous media and in vitro (HeLa cells), which confirms that the guests can also be exchanged intracellularly.
Two mechanisms for guest transfer between hosts are proposed: 1) guests escape a host and subsequently are captured by another or 2) collision between hosts leads to exchange of guests. They indicate that because of the poor aqueous solubility of the fluorophore guests, mechanism 2 is more likely, but the authors do not perform any experiments to confirm this. They also state that 10-fold dilution with PBS does not affect energy-transfer efficiency. This is not expected if mechanism 2 is at play. Dilution would lead to less frequent collisions between hosts, and therefore, to less guest exchange, and less energy-transfer. The energy-transfer observed could be explained by diffusion, which is more akin to mechanism 1. Poor solubility might not be an issue, because as long as a small amount is soluble, escapes the host and is further entrapped by a host carrying the opposite guest, energy-transfer can still occur.
The authors attempt to determine where the supramolecular assemblies are localized intracellularly by incubating HeLa cells with two sets of nanoparticles, loaded separately with anthracene and BODIPY, and either chlorpromazine or genistein, which inhibit either clathrin- or caveolae-mediated endocytosis, respectively. The results indicated that intracellular fluorescence was reduced to a greater extent by the addition of chlorpromazine, which suggests that the particles are predominantly internalized by clathrin-mediated endocytosis. However, the authors do not perform further experiments that show the particles localized in endosomes and/or lysosomes, which would result from clathrin-mediated endocytosis. They seem to be attempting to rule out any other methods of internalization, but given that chlorpromazine only reduced the intracellular fluorescence to 59%, other methods of internalization could still be taking place.
Swaminathan, 2014. Intracellular Guest Exchange between Dynamic Supramolecular Hosts
Carla’s comment on: Active Targeting of the Nucleus Using Nonpeptidic Boronate Tags
By Carla M. Quiñones
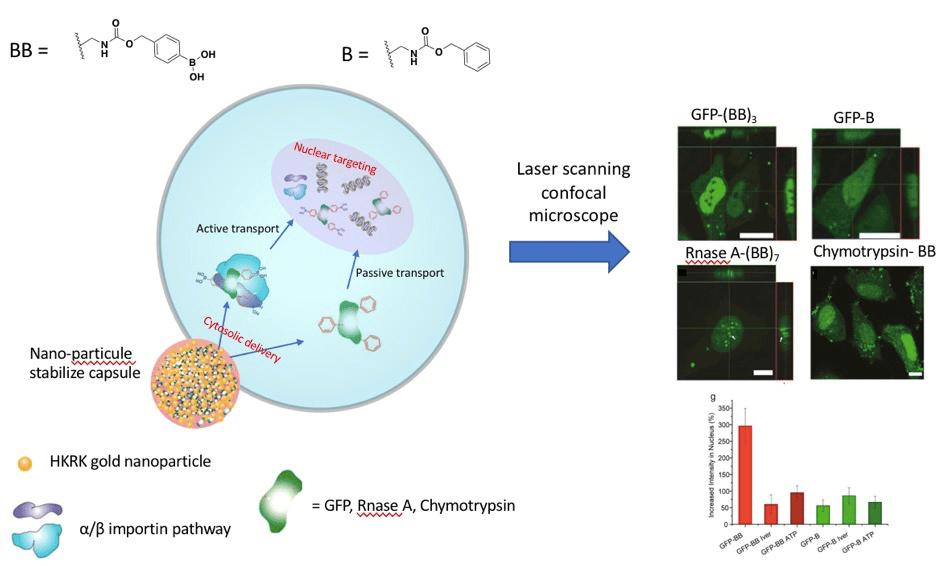
The main objective of the research described in this article by Rotello and coworkers was to use a synthetic non-peptide targeting motif that accesses the nucleus of the cell through an active transport mechanism. Active and passive transport take place in the cellular membrane as well as in the nuclear membrane. Intracellular targeting is as important as cellular targeting due to their relevance in drug delivery and potential therapies. The two challenges faced by the research group was to deliver the nuclear-targeted protein into the cytosol and to comprehend the mode of nuclear entry. They delivered the protein successfully to the cytosol with the help of a previously synthesised nanoparticle stabilized particle, which encapsulated the proteins of interest and liberated them into the cytosol via membrane fusion. The most significant finding was the efficiency of targeting the nucleus when modifying each protein of interest with benzyl boronate tags (BB tags).
They performed experiments with different proteins modified with BB tags resulting in a successful high-efficiency delivery to the nucleus. To assess the role of boronic acid in the BB tags, they modified GFP with the benzyl tag alone and saw less fluorescence inside the nucleus. This suggests that the boronic acid is necessary in the BB tags for a successful nuclear targeting. Furthermore, they determined if the mechanism of transport into the nucleus was either active or passive. To accomplish this goal, they added Ivermectin, an inhibitor for the α/β importin pathway (active transport), to the cells and also depleted ATP (required for all active transport pathways) in another set of experiments. They delivered GFP with 3 BB tags in both cases and saw a lower nuclear efficiency. They concluded that these modified proteins targeted the nucleus effectively through the importin α/β pathway (active transport) rather than passive transport.
Through the last part of the paper, I was curious of how they acknowledged that this happened the way the proposed since they didn’t explain the mechanism behind the α/β importin pathway, nor the chemistry of how the BB tag contributes to the nuclear targeting. Intrigued by this, I searched more about this phenomenon, but found no relevant studies, probably because this is a novel research field. In general, this was a good article and useful to our lab since we work with supramolecular systems which, in the future, we could adapt this strategy to achieve nuclear targeting.