Tag: copolymers
Minelise’s comment on: Synthesis and Direct Observation of Thermoresponsive DNA Copolymers
By Minelise E. Rivera
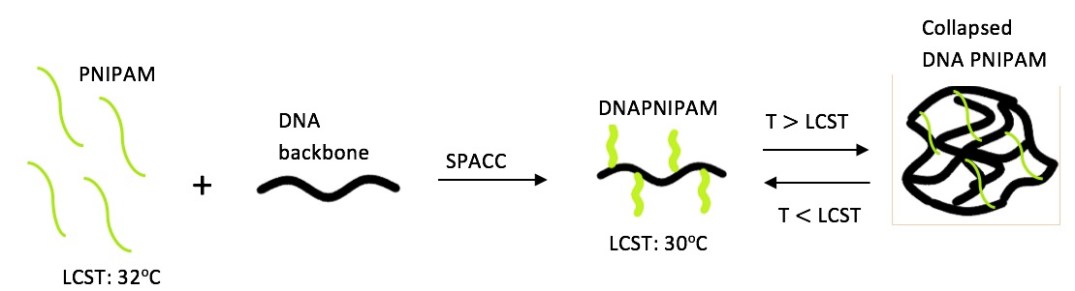
In this paper, Li and Schroeder use single molecule techniques to have a direct observation on DNA-PNIPAM copolymers. First, they synthetized DNA-PNIPAM copolymers using a two-step strategy based on polymerase chain reaction (PCR) for generating linear DNA backbones containing dibenzocyclooctyne-dUTP, then grafted thermoresponsive side branches (PNIPAM) onto DNA backbones using copper-free click chemistry. Subsequent single molecule fluorescence spectroscopy studies unveiled more clearly the molecular heterogeneity association with the stretching and relaxing of the polymer above and below their LCST. Their results showed that intramolecular conformational dynamics of DNA-PNIPAM copolymers are affected by properties of the branches like molecular weight, density, leading to a change in transition temperatures. In other words, the single molecule technique provided a better understanding in a molecular perspective of chemically heterogeneous and stimuli-response polymers.
As I read this paper and looked for information to better understand it, I was amazed by the details with which they worked with throughout their study. I would have thought of working better with a bunch of them instead of just single molecules. It didn’t cross my mind that someone was going to, not only synthesized the molecule, but also study its characteristics. It was very interesting to learn about the methods that they used for characterization and synthesis. It got me wondering if those methods were the only ones that would work in this case and why. But, what I think that was very useful for me is that I got to understand better the importance of the LCST and the role that it played in their system. It reminded me of our project in which the SGQ self-assembles into the SHS and how it is to study it and understand its influence in the SHS as it was important for the copolymers with which it was worked with in the paper.
Valeria’s comment on: Reversible Regulation of Thermoresponsive Property of Dithiomaleimide-Containing Copolymers via Sequential Thiol Exchange Reactions
By Valeria Burgos Caldero
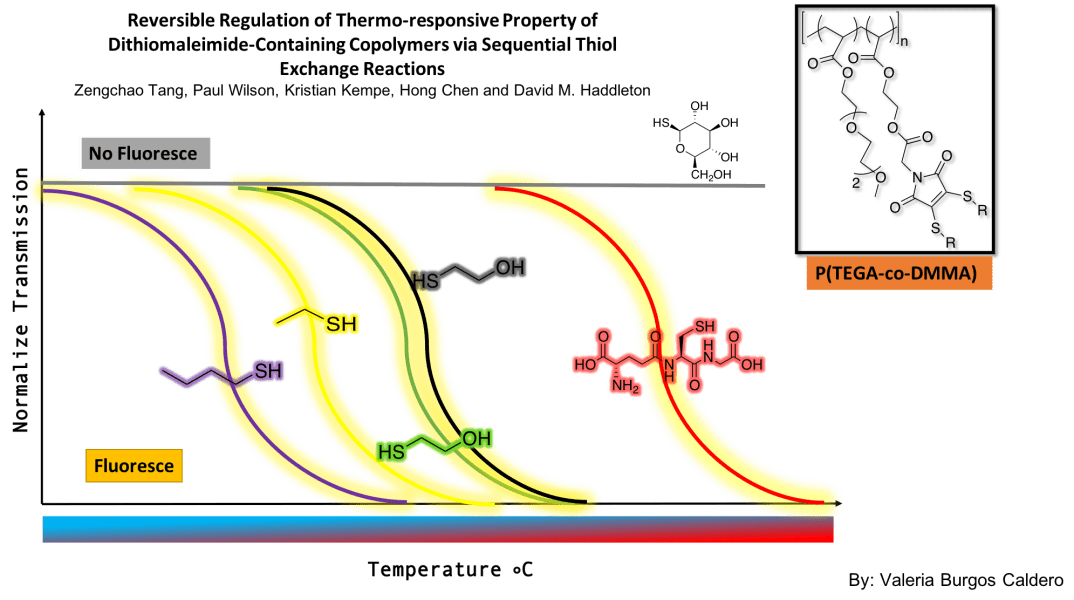
The main purpose of this article was to synthesize multi-responsive polymers that could be reversibly modified to adjust their LCST. Indeed, these researchers were able to develop a system in which multiple thiol exchanges were made, and in turn, they could determine how the thiols affected the transition temperature of the polymer. They used a copolymer containing P(TEGA) and DMMA. By performing transmission measurements at various temperatures, they concluded that as the thiol changed, the transition temperature of polymer varied depending on the resulting hydrophobicity. More polar functionalities increased the transition temperature and less polar ones decreased it. They were able to demonstrate the reversibility of the modifications since they managed to return to their original functionality after various thiol exchanges. Finally, they implemented a fluorescence signal to monitor the reaction progress. They found that thioglucose quenches the polymer’s fluorescence while making the compound soluble throughout the range of temperatures. With these findings, a wide range of possibilities were opened, since now, if you want a polymer for a specific type of function where a specific temperature response is needed, it is easily accessible by adding the corresponding thiol to the polymer solution. The mechanism of turning off the fluorescence may give access to reversible systems in aqueous conditions.
In general, I found it much simpler to prepare for this article than for the first one I presented. I feel that by doing these exercises of presenting scientific articles I have been acquiring maturity in the analysis process since it was difficult for me to understand articles in the beginning. Something that I found missing in the article is that they never explained the experimental procedure on how they achieved reversibility after adding different thiols to the same sample. I liked that they used common thiols, some of which we use in our research and others that maybe we could apply. In general, the article relates a lot to the research I’m doing with Diana. It could be useful to try to see the stimuli-responsive variations in the compounds that we are synthesizing. Maybe because it is related to variations in functionalities with thiols, similar to my own research, I found it more enjoyable to prepare the discussion and understand the material in the article.