Tag: polymers
Rafa’s comment on: Thermal Switching of Thermoresponsive Polymer Aqueous Solutions
By Rafael A. Brito
Thermal switches are of great importance to thermal management in a wide variety of applications. A common characteristic associated with thermal switching is thermal conductivity. After noticing the change in thermal conductivity across LCST transitions, Zhiting Tian started researching polymers for this purpose. Poly(N-isopropylacrylamide) (PNIPAM) is the most studied thermoresponsive polymer and has a workable LCST of 32° C. The LCST transition of PNIPAM changes the chain-like formation of the polymer into an aggregation that shows a drastic decrease of thermal conductivity. This sharp change is due to LCST transitions being second-order, which are characterized by being almost instantaneous when the corresponding temperature is reached. Thermal conductivity was measured by applying a powerful approach: the transient thermal grating technique. It is used by heating a solution as a function of position creating a grating of temperature. This grating allows the use of the one-dimensional heat equation, which can be solved to give a relation between the thermal conductivity and temperature. The thermal conductivity can then be calculated using 𝑘 = 𝜌𝑐𝑝α, where 𝜌 is the density, is the specific heat capacity and α which is a function of temperature. After the setup was completed, solutions of varying concentrations were analyzed. For the solution with the highest concentration, the thermal switching ratio was measured to be 1.15 across the LCST transition. This shows a significant change between the two states of the polymer. The thermal conductivity of the PNIPAM aqueous solutions increases with temperature, the same as with water, until reaching the LCST. Then a drastic change is observed in the solution. The thermal switching ratio of PNIPAM aqueous solutions across the transition keeps increasing with increasing concentration, which is expected from the equation. To explain the thermal conductivity change due to the transition between the two modes of the polymer, the authors used the idea that the homogeneous phase of the solution separates into two phases that increases the thermal interface resistance resulting in a lower effective thermal conductivity.
As a summary, they reported the first direct measurement of thermal conductivity change in PNIPAM aqueous solutions across the LCST using a powerful approach, the laser-induced transient thermal grating technique. The results show an abrupt thermal conductivity drop across the transition temperature. The potential of using thermoresponsive polymer aqueous solutions of higher-order phase transitions for thermal switch applications has been demonstrated throughout this paper’s work.
Maxier’s comment on: Macromolecular Crowding Modifies the Impact of Specific Hofmeister Ions on the Coil–Globule Transition of PNIPAM
By Maxier Acosta Santiago
While the recently discussed Cremer paper described efforts towards understanding the effect on the LCST of PNIPAM by salts of the Hofmeister’s series, Sakota’s article went somewhat deeper into the study of this phenomenon by taking in consideration molecular crowding. Sakota’s group decided to use a PEG polymer as a crowding agent. Crowding may affect the biding of anions from the Hofmeister’s series and the PNIPAM resulting in a change in the LCST. First, they studied the presence of PEG in a PNIPAM solution showing that the crowding agent reduces the LCST. Kosmotropic anions, that decrease the LCST, but chaotropes increase the LCST. For the Hofemeister series effect on LCST we can go back to Luis Prieto’s blog post and Cremer’s paper which explains this effect better.
When the article begins to look at the presence of PEG at different salt concentrations, they see a close correlation between the LCST and the Hofmeister series. Yet, for the chaotropes ClO4⁻ and SCN⁻, the presence of PEG lead to a larger increase in the LCST. From here they decide to apply different theories to explain the results. Within the examined theories they discuss around thermodynamics of the system. Their explanation evolves as follows, even if the organization via LCST of PNIPAM is not thermodynamically favorable, the overlapping excluded volumes of PEG and the PNIPAM particles increase the translational entropy of water molecules in solution, which makes the formation of the system possible.
Although this paper brings something new to the table to discuss (molecular crowding and LCST), I do have some concerns. When taking into consideration so many different factors like molecular crowding, salt, and the responsive system itself, we should look deeper into the behavior. To limit certain factors, they maintained certain constant concentrations throughout the paper. Yet, in the discussion, I feel they lacked additional experimental results or computational studies (using molecular and/or coarse-grained simulations) to support their theoretical thermodynamics analysis.
Minelise’s comment on: Synthesis and Direct Observation of Thermoresponsive DNA Copolymers
By Minelise E. Rivera
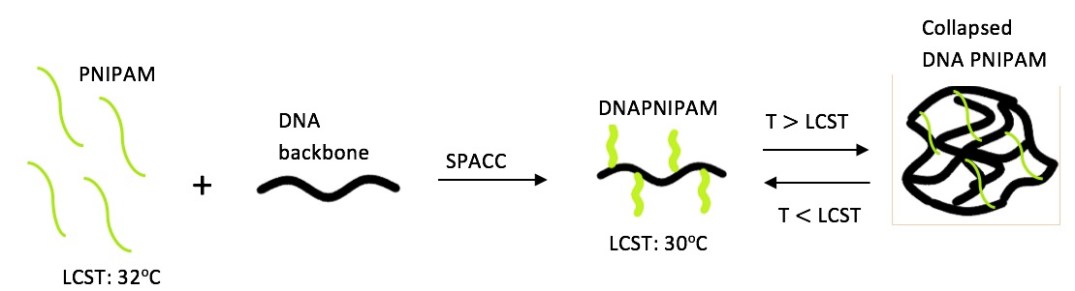
In this paper, Li and Schroeder use single molecule techniques to have a direct observation on DNA-PNIPAM copolymers. First, they synthetized DNA-PNIPAM copolymers using a two-step strategy based on polymerase chain reaction (PCR) for generating linear DNA backbones containing dibenzocyclooctyne-dUTP, then grafted thermoresponsive side branches (PNIPAM) onto DNA backbones using copper-free click chemistry. Subsequent single molecule fluorescence spectroscopy studies unveiled more clearly the molecular heterogeneity association with the stretching and relaxing of the polymer above and below their LCST. Their results showed that intramolecular conformational dynamics of DNA-PNIPAM copolymers are affected by properties of the branches like molecular weight, density, leading to a change in transition temperatures. In other words, the single molecule technique provided a better understanding in a molecular perspective of chemically heterogeneous and stimuli-response polymers.
As I read this paper and looked for information to better understand it, I was amazed by the details with which they worked with throughout their study. I would have thought of working better with a bunch of them instead of just single molecules. It didn’t cross my mind that someone was going to, not only synthesized the molecule, but also study its characteristics. It was very interesting to learn about the methods that they used for characterization and synthesis. It got me wondering if those methods were the only ones that would work in this case and why. But, what I think that was very useful for me is that I got to understand better the importance of the LCST and the role that it played in their system. It reminded me of our project in which the SGQ self-assembles into the SHS and how it is to study it and understand its influence in the SHS as it was important for the copolymers with which it was worked with in the paper.
José’s comment on: Intelligent, Biodegradable, and Self-Healing Hydrogels Utilizing DNA Quadruplexes
By José M. Martinez
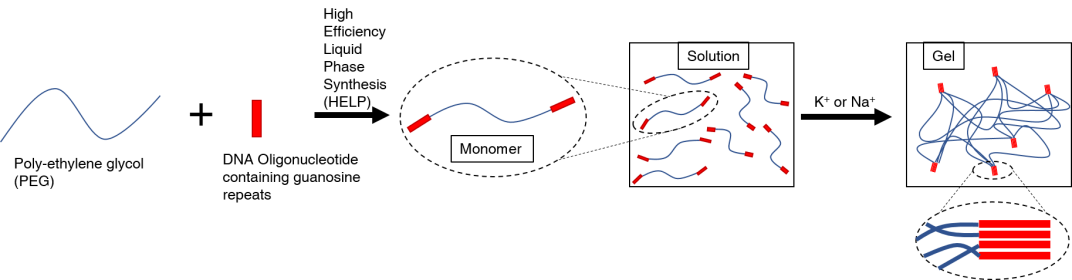
This paper is about making of a water based gel made from the supramolecular interactions of guanosine rich oligonucleotides. They attached these oligonucleotides symmetrically into the ends of PEG chains of variable lengths and conformations. This was made through High Efficiency Liquid Phase (HELP) synthesis. This synthetic strategy, as its name says, occurs in a liquid phase instead of the usual solid phase synthesis. The purpose of this is to allow them to obtain greater quantities of product more easily without risking the effectiveness. These DNA-PEG-DNA monomers have the ability, when diluted and presented with a cation (K+ or Na+), to self-assemble its DNA moieties through non-covalent interactions to for G-quadruplexes. Those strong interactions change the physical properties of the solution by forming a hydrogel. In the study, it was proven that the concentration of the cation required to trigger the self-assembly is relatively small. It is even compared to the concentration of salts found in body-related fluids like sweat, saliva, and tears. The hydrogel also has the capability to heal itself and interact by diffusion with another of the same kind. Finally, it was found that by adding a few extra nucleotides to the DNA moiety of the monomer and a matching strand for that part, the G-quadruplexes could be disassembled, returning the gel back to its liquid phase. All the previously stated properties indicate potential uses of this hydrogel for biomedical purposes.
Regarding myself, I learned mostly about the molecular aspect of gels and how they are formed. It was very interesting seeing the role that the supramolecular interactions played in the formation of this particular gel and the experiments made to investigate further into its properties.
JMM: Tanaka, 2017. Intelligent, Biodegradable, and Self-Healing Hydrogels Utilizing DNA Quadruplexes
Valeria’s comment on: Reversible Regulation of Thermoresponsive Property of Dithiomaleimide-Containing Copolymers via Sequential Thiol Exchange Reactions
By Valeria Burgos Caldero
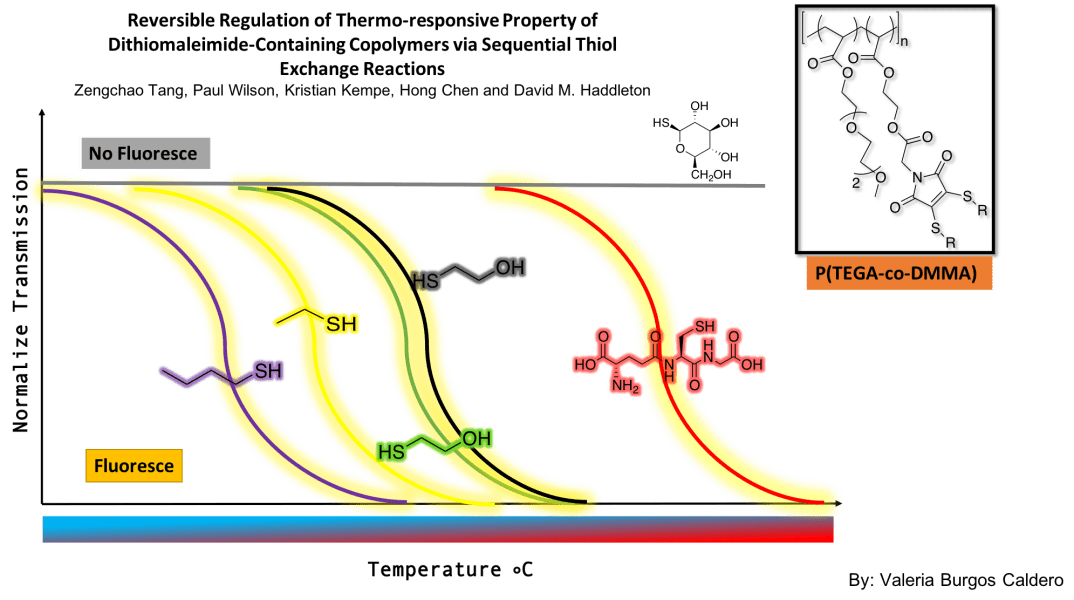
The main purpose of this article was to synthesize multi-responsive polymers that could be reversibly modified to adjust their LCST. Indeed, these researchers were able to develop a system in which multiple thiol exchanges were made, and in turn, they could determine how the thiols affected the transition temperature of the polymer. They used a copolymer containing P(TEGA) and DMMA. By performing transmission measurements at various temperatures, they concluded that as the thiol changed, the transition temperature of polymer varied depending on the resulting hydrophobicity. More polar functionalities increased the transition temperature and less polar ones decreased it. They were able to demonstrate the reversibility of the modifications since they managed to return to their original functionality after various thiol exchanges. Finally, they implemented a fluorescence signal to monitor the reaction progress. They found that thioglucose quenches the polymer’s fluorescence while making the compound soluble throughout the range of temperatures. With these findings, a wide range of possibilities were opened, since now, if you want a polymer for a specific type of function where a specific temperature response is needed, it is easily accessible by adding the corresponding thiol to the polymer solution. The mechanism of turning off the fluorescence may give access to reversible systems in aqueous conditions.
In general, I found it much simpler to prepare for this article than for the first one I presented. I feel that by doing these exercises of presenting scientific articles I have been acquiring maturity in the analysis process since it was difficult for me to understand articles in the beginning. Something that I found missing in the article is that they never explained the experimental procedure on how they achieved reversibility after adding different thiols to the same sample. I liked that they used common thiols, some of which we use in our research and others that maybe we could apply. In general, the article relates a lot to the research I’m doing with Diana. It could be useful to try to see the stimuli-responsive variations in the compounds that we are synthesizing. Maybe because it is related to variations in functionalities with thiols, similar to my own research, I found it more enjoyable to prepare the discussion and understand the material in the article.