By José M. Martinez
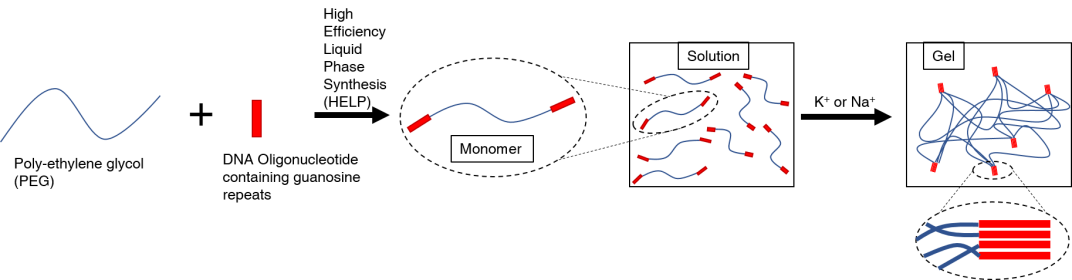
This paper is about making of a water based gel made from the supramolecular interactions of guanosine rich oligonucleotides. They attached these oligonucleotides symmetrically into the ends of PEG chains of variable lengths and conformations. This was made through High Efficiency Liquid Phase (HELP) synthesis. This synthetic strategy, as its name says, occurs in a liquid phase instead of the usual solid phase synthesis. The purpose of this is to allow them to obtain greater quantities of product more easily without risking the effectiveness. These DNA-PEG-DNA monomers have the ability, when diluted and presented with a cation (K+ or Na+), to self-assemble its DNA moieties through non-covalent interactions to for G-quadruplexes. Those strong interactions change the physical properties of the solution by forming a hydrogel. In the study, it was proven that the concentration of the cation required to trigger the self-assembly is relatively small. It is even compared to the concentration of salts found in body-related fluids like sweat, saliva, and tears. The hydrogel also has the capability to heal itself and interact by diffusion with another of the same kind. Finally, it was found that by adding a few extra nucleotides to the DNA moiety of the monomer and a matching strand for that part, the G-quadruplexes could be disassembled, returning the gel back to its liquid phase. All the previously stated properties indicate potential uses of this hydrogel for biomedical purposes.
Regarding myself, I learned mostly about the molecular aspect of gels and how they are formed. It was very interesting seeing the role that the supramolecular interactions played in the formation of this particular gel and the experiments made to investigate further into its properties.
JMM: Tanaka, 2017. Intelligent, Biodegradable, and Self-Healing Hydrogels Utilizing DNA Quadruplexes