Tag: thermoresponsive
Rafa’s comment on: Thermal Switching of Thermoresponsive Polymer Aqueous Solutions
By Rafael A. Brito
Thermal switches are of great importance to thermal management in a wide variety of applications. A common characteristic associated with thermal switching is thermal conductivity. After noticing the change in thermal conductivity across LCST transitions, Zhiting Tian started researching polymers for this purpose. Poly(N-isopropylacrylamide) (PNIPAM) is the most studied thermoresponsive polymer and has a workable LCST of 32° C. The LCST transition of PNIPAM changes the chain-like formation of the polymer into an aggregation that shows a drastic decrease of thermal conductivity. This sharp change is due to LCST transitions being second-order, which are characterized by being almost instantaneous when the corresponding temperature is reached. Thermal conductivity was measured by applying a powerful approach: the transient thermal grating technique. It is used by heating a solution as a function of position creating a grating of temperature. This grating allows the use of the one-dimensional heat equation, which can be solved to give a relation between the thermal conductivity and temperature. The thermal conductivity can then be calculated using 𝑘 = 𝜌𝑐𝑝α, where 𝜌 is the density, is the specific heat capacity and α which is a function of temperature. After the setup was completed, solutions of varying concentrations were analyzed. For the solution with the highest concentration, the thermal switching ratio was measured to be 1.15 across the LCST transition. This shows a significant change between the two states of the polymer. The thermal conductivity of the PNIPAM aqueous solutions increases with temperature, the same as with water, until reaching the LCST. Then a drastic change is observed in the solution. The thermal switching ratio of PNIPAM aqueous solutions across the transition keeps increasing with increasing concentration, which is expected from the equation. To explain the thermal conductivity change due to the transition between the two modes of the polymer, the authors used the idea that the homogeneous phase of the solution separates into two phases that increases the thermal interface resistance resulting in a lower effective thermal conductivity.
As a summary, they reported the first direct measurement of thermal conductivity change in PNIPAM aqueous solutions across the LCST using a powerful approach, the laser-induced transient thermal grating technique. The results show an abrupt thermal conductivity drop across the transition temperature. The potential of using thermoresponsive polymer aqueous solutions of higher-order phase transitions for thermal switch applications has been demonstrated throughout this paper’s work.
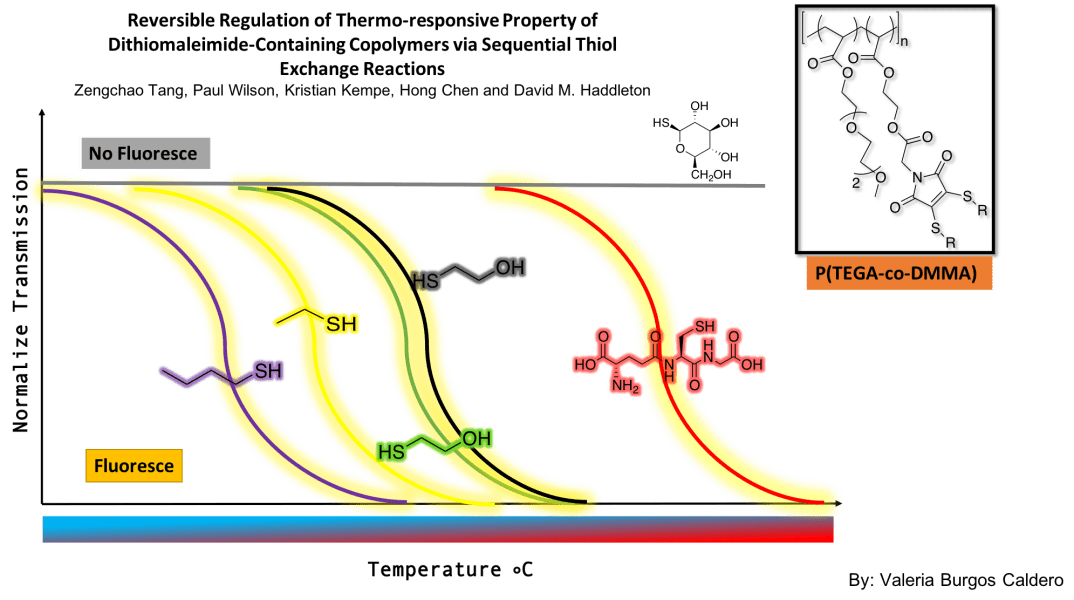
Valeria’s comment on: Reversible Regulation of Thermoresponsive Property of Dithiomaleimide-Containing Copolymers via Sequential Thiol Exchange Reactions
By Valeria Burgos Caldero
The main purpose of this article was to synthesize multi-responsive polymers that could be reversibly modified to adjust their LCST. Indeed, these researchers were able to develop a system in which multiple thiol exchanges were made, and in turn, they could determine how the thiols affected the transition temperature of the polymer. They used a copolymer containing P(TEGA) and DMMA. By performing transmission measurements at various temperatures, they concluded that as the thiol changed, the transition temperature of polymer varied depending on the resulting hydrophobicity. More polar functionalities increased the transition temperature and less polar ones decreased it. They were able to demonstrate the reversibility of the modifications since they managed to return to their original functionality after various thiol exchanges. Finally, they implemented a fluorescence signal to monitor the reaction progress. They found that thioglucose quenches the polymer’s fluorescence while making the compound soluble throughout the range of temperatures. With these findings, a wide range of possibilities were opened, since now, if you want a polymer for a specific type of function where a specific temperature response is needed, it is easily accessible by adding the corresponding thiol to the polymer solution. The mechanism of turning off the fluorescence may give access to reversible systems in aqueous conditions.
In general, I found it much simpler to prepare for this article than for the first one I presented. I feel that by doing these exercises of presenting scientific articles I have been acquiring maturity in the analysis process since it was difficult for me to understand articles in the beginning. Something that I found missing in the article is that they never explained the experimental procedure on how they achieved reversibility after adding different thiols to the same sample. I liked that they used common thiols, some of which we use in our research and others that maybe we could apply. In general, the article relates a lot to the research I’m doing with Diana. It could be useful to try to see the stimuli-responsive variations in the compounds that we are synthesizing. Maybe because it is related to variations in functionalities with thiols, similar to my own research, I found it more enjoyable to prepare the discussion and understand the material in the article.
Our last three papers…
 Below are the references to our last three papers. I will post a brief overview of each one soon, but in the meantime:
Below are the references to our last three papers. I will post a brief overview of each one soon, but in the meantime:
Structural studies of supramolecular G-quadruplexes formed from 8-aryl-2’-deoxyguanosine derivatives. García-Arriaga, M.; Hobley, G.; Rivera, J. M., J. Org. Chem.2016, 81, Advance Online Publication; DOI: 10.1021/acs.joc.6b01113. PMID: 27303787
- The first 50 people can download a free reprint of the paper directly from the publisher by going to this link.
- Abstract. Self-assembly is a powerful tool for the construction of complex nanostructures. Despite the advances in the field, the development of precise self-assembled structures remains a challenge. We have shown that in the presence of suitably sized cations like K+, 8-aryl-2′-deoxyguanosine (8ArG) derivatives self-assemble into sets of coaxially stacked planar tetramers, we term supramolecular G-quadruplexes (SGQs). Previously, we reported that when the 8-aryl group is a phenyl ring with a meta-carbonyl group, the resulting supramolecule is a hexadecamer, which is remarkably robust as illustrated by its isostructural assembly in both organic and aqueous environments. We report here a detailed three-dimensional structure of the SGQs formed by lipophilic, and hydrophilic, 8ArG derivatives with either 8-(meta-acetylphenyl), 8-(para-acetylphenyl), and 8-(meta-ethoxycarbonylphenyl) groups. The chirality and close contacts between the subunits impose different levels of steric and electrostatic constraints on opposite sides of the tetrads, which determine their preferred relative orientation. The balance between attractive non-covalent interactions juxtaposed with repulsive steric and electrostatic interactions explains the high cooperativity, fidelity and stability of these SGQs. These structural studies, together with titration experiments and molecular dynamics simulations provide insight on the mechanism of formation of these SGQs.
Organic Nanoflowers From a Wide Variety of Molecules Templated By A Hierarchical Supramolecular Scaffold. Negrón, L. M.; Diaz, T. L.; Ortiz-Quiles, E. O.; Dieppa, D.; Madera-Soto, B.; Rivera, J. M., Langmuir 2016, 32 (10), 2283–2290. DOI: 10.1021/acs.langmuir.5b03946; PMCID: PMC4896646
- Abstract. Nanoflowers (NFs) are flowered-shaped particles with overall sizes or features in the nanoscale. Beyond their pleasing aesthetics, NFs have found a number of applications ranging from catalysis, to sensing, to drug delivery. Compared to inorganic based NFs, their organic and hybrid counterparts are relatively underdeveloped mostly because of the lack of a reliable and versatile method for their construction. We report here a method for constructing NFs from a wide variety of biologically relevant molecules (guests), ranging from small molecules, like doxorubicin, to biomacromolecules, like various proteins and plasmid DNA. The method relies on the encapsulation of the guests within a hierarchically structured particle made from supramolecular G-quadruplexes. The size and overall flexibility of the guests dictate the broad morphological features of the resulting NFs, specifically, small and rigid guests favor the formation of NFs with spiky petals, while large and/or flexible guests promote NFs with wide petals. The results from experiments using confocal fluorescence microscopy, and scanning electron microscopy provides the basis for the proposed mechanism for the NF formation.
Tuning Thermoresponsive Supramolecular G-Quadruplexes. José E. Betancourt & José M. Rivera, Langmuir 2015, 31 (7), 2095-2103. DOI:10.1021/la504446k; PMCID: PMC4863471 [Free PMC Article]
- Abstract. Thermoresponsive systems are attractive due to their suitability for fundamental studies as well as their practical uses in a wide variety of applications. While much progress has been achieved using polymers, alternative strategies such as the use of well-defined nonpolymeric supramolecules are still underdeveloped. Here we report three 8-aryl-2′-deoxyguanosine derivatives (8ArGs) that self-assemble in aqueous media into precise thermoresponsive supramolecular G-quadruplexes (SGQs). We report the synthesis of such derivatives, studies of their isothermal self-assembly, and the thermally induced assembly to form higher-order meso-globular assemblies we term supramolecular hacky sacks (SHS). The lower critical solution temperature (LCST) that indicates the formation of the SHS was modulated by changing (a) intrinsic parameters (i.e., structure of the 8ArGs); (b) extrinsic parameters such as the salt used to promote the formation of the SGQ; and (c) supramolecular parameters such as the coassembly different 8ArGs to form heteromeric SGQs. Changes in the intrinsic parameters lead to LCST variations in the range of 28–59 °C. Modulating extrinsic parameters such as replacing KI with KSCN abolishes the thermoresponsive phenomenon whereas changing the cation from K+ to Na+or adjusting the pH (in the range of 6–8) has negligible effects on the LCST. Modulating supramolecular parameters results in transition temperatures that are intermediate between those obtained by the respective homomeric SGQs, although the specific proportions of the subunits are critical in determining the reversibility of the process. Given the extensive applications of thermoresponsive polymers, the nonpolymeric supramolecular counterparts presented here may represent an attractive alternative for fundamental studies and biorelevant applications.