By Yanira Rodríguez Valdéz
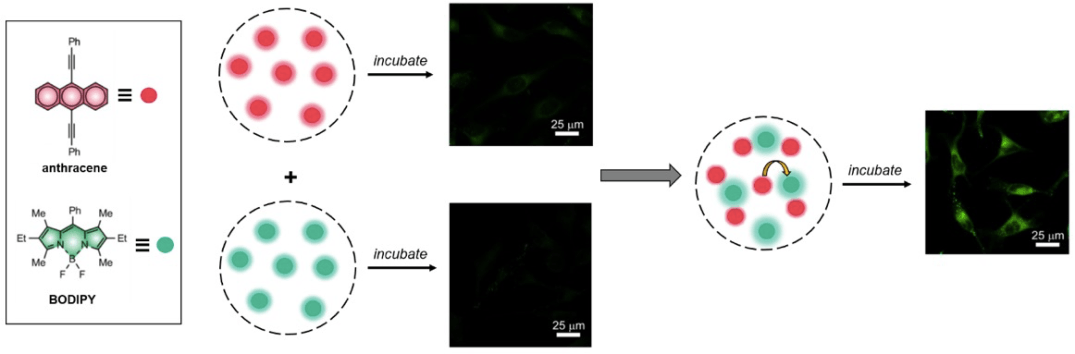
The article by Swaminathan et al. studies the Förster Resonance Energy Transfer (FRET) between two dyes encapsulated in self-assembled nanoparticles made from amphiphilic polymers. The encapsulation of hydrophobic chromophores such as borondipyrromethene (BODIPY) and anthracene inside the hydrophobic interior of these nanoparticles allows their study in aqueous media. In close proximity (inside the same host), BODIPY and anthracene undergo FRET (anthracene = donor, BODIPY = acceptor). The authors observed that mixing nanoparticles loaded with anthracene and nanoparticles loaded with BODIPY has the same effect as loading nanoparticles with both anthracene and BODIPY, which suggests guests being exchanged when nanoparticles are mixed and that they are being captured by the same nanoparticle(s). These studies are performed both in aqueous media and in vitro (HeLa cells), which confirms that the guests can also be exchanged intracellularly.
Two mechanisms for guest transfer between hosts are proposed: 1) guests escape a host and subsequently are captured by another or 2) collision between hosts leads to exchange of guests. They indicate that because of the poor aqueous solubility of the fluorophore guests, mechanism 2 is more likely, but the authors do not perform any experiments to confirm this. They also state that 10-fold dilution with PBS does not affect energy-transfer efficiency. This is not expected if mechanism 2 is at play. Dilution would lead to less frequent collisions between hosts, and therefore, to less guest exchange, and less energy-transfer. The energy-transfer observed could be explained by diffusion, which is more akin to mechanism 1. Poor solubility might not be an issue, because as long as a small amount is soluble, escapes the host and is further entrapped by a host carrying the opposite guest, energy-transfer can still occur.
The authors attempt to determine where the supramolecular assemblies are localized intracellularly by incubating HeLa cells with two sets of nanoparticles, loaded separately with anthracene and BODIPY, and either chlorpromazine or genistein, which inhibit either clathrin- or caveolae-mediated endocytosis, respectively. The results indicated that intracellular fluorescence was reduced to a greater extent by the addition of chlorpromazine, which suggests that the particles are predominantly internalized by clathrin-mediated endocytosis. However, the authors do not perform further experiments that show the particles localized in endosomes and/or lysosomes, which would result from clathrin-mediated endocytosis. They seem to be attempting to rule out any other methods of internalization, but given that chlorpromazine only reduced the intracellular fluorescence to 59%, other methods of internalization could still be taking place.
Swaminathan, 2014. Intracellular Guest Exchange between Dynamic Supramolecular Hosts